Navigating the Path to GRAS Notice: A Comprehensive Guide to Ensuring Food Safety in Canada and the USA

In the dynamic and ever-evolving landscape of the food industry, consumer safety stands as a non-negotiable priority. A cornerstone in this endeavor is securing the Generally Recognized as Safe (GRAS) notice, a designation recognized by both the U.S. Food and Drug Administration (FDA) and Health Canada. GRAS notice is more than just a regulatory hurdle; it’s a testament to a company’s unwavering commitment to delivering safe and top-quality food products. Embark on a comprehensive journey with us to unravel the significance, intricacies, and impact of GRAS notice in both Canada and the USA.
Understanding the Significance of GRAS Notice
GRAS notice plays a pivotal role in the food industry, serving as a critical benchmark for food manufacturers seeking to market their products in Canada and the USA. This designation signifies that the FDA or Health Canada, respectively, has meticulously evaluated the scientific data and concluded that the food ingredient is safe for human consumption under the intended conditions of use. This designation instils confidence in consumers, assuring them that the products they purchase meet the highest safety standards.
Benefits of GRAS Certification
Beyond ensuring regulatory compliance, GRAS notice offers a multitude of benefits for food manufacturers, including:
- Enhanced Brand Reputation: GRAS notice serves as a badge of honor, demonstrating a company’s unwavering commitment to food safety and quality. It fosters consumer trust, leading to increased brand loyalty, market recognition, and a competitive edge.
- Expanded Market Access: GRAS notice opens doors to new markets, allowing manufacturers to expand their product reach and revenue potential. It facilitates the export of products to countries that recognize GRAS status, paving the way for global expansion.
- Reduced Regulatory Hurdles: GRAS notice streamlines the regulatory process, minimizing the time and resources required to gain market approval. It eliminates the need for pre-market approval for food ingredients that have already been deemed safe, streamlining the process for manufacturers.
- Enhanced Consumer Confidence: GRAS notice serves as a visible assurance to consumers that the products they purchase have been thoroughly evaluated and deemed safe for consumption. This fosters trust and encourages repeat purchases.
Step-by-Step Guide to GRAS Certification
The GRAS notice process is rigorous and demands meticulous preparation. Here’s a comprehensive step-by-step guide:
1. Conduct a Thorough Safety Assessment:
Before submitting a GRAS notice, manufacturers must undertake a comprehensive safety assessment of their food ingredient. This assessment should consider all potential risks, including toxicity, dietary exposure, and age limitations. The assessment should be based on sound scientific principles and be supported by robust data.
2. Identify the Appropriate Notice Type:
There are two types of GRAS notices: GRAS and GRAS Affirmed. A GRAS notice signifies the manufacturer’s belief that the ingredient is GRAS, while a GRAS Affirmed notice formally requests the FDA or Health Canada’s affirmation of the ingredient’s safety. The choice of notice type depends on the intended use of the ingredient and the strength of the supporting evidence.
3. Prepare a Comprehensive GRAS Dossier:
The GRAS dossier is the cornerstone of the GRAS notice process. It should include detailed information on the ingredient’s identity, manufacturing methods, dietary exposure, historical use, and supporting data. The dossier should be meticulously compiled to ensure it provides a comprehensive overview of the ingredient’s safety profile.
4. Submit the GRAS Notice and Engage with Regulatory Authorities:
Once the GRAS dossier is complete, it must be submitted to the appropriate regulatory agency, either the FDA or Health Canada. Active engagement with the agency throughout the review process is crucial for addressing any questions or concerns promptly. This open communication ensures that the agency’s feedback is incorporated and that the notice process proceeds smoothly.
5. Maintain Ongoing Safety Monitoring:
Obtaining GRAS notice is not the end of the journey. Manufacturers must continuously monitor the safety of their GRAS ingredients and adhere to all applicable regulations. This includes staying up-to-date on scientific developments, conducting post-market surveillance, and promptly addressing any potential safety concerns. Ongoing vigilance is essential to maintain the safety of products and uphold the reputation of the company.
To assist companies in obtaining GRAS notice, various services are available. These include feasibility reviews, identification of evidence gaps, organization of toxicological studies, preparation of GRAS notification dossiers, engagement of expert panel members, FDA filing, and facilitation of meetings with the FDA and Health Canada. These services streamline the process, ensuring that companies meet all necessary requirements for certification. If you want to learn more about GRAS Certification, get in touch with experts at Quality Smart Solutions.

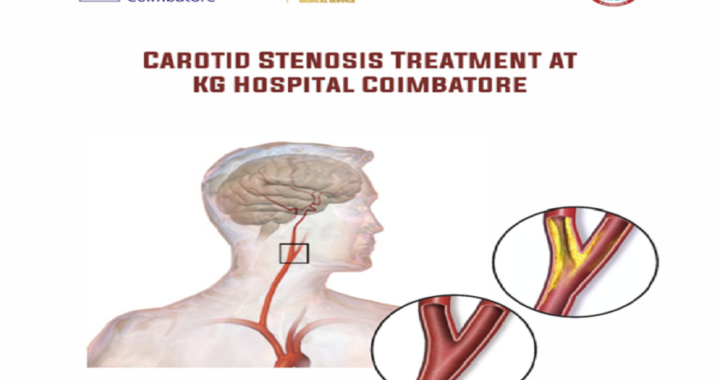 Carotid stenosis
Carotid stenosis  The Aesthetic and Functional Benefits of Compression Gear
The Aesthetic and Functional Benefits of Compression Gear 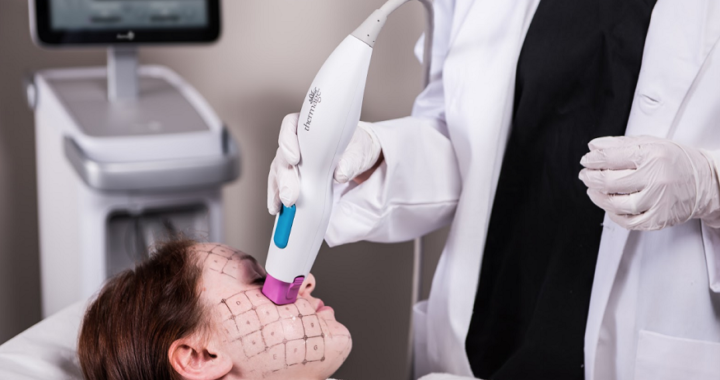 Why Is Thermage FLX So Expensive in Singapore
Why Is Thermage FLX So Expensive in Singapore 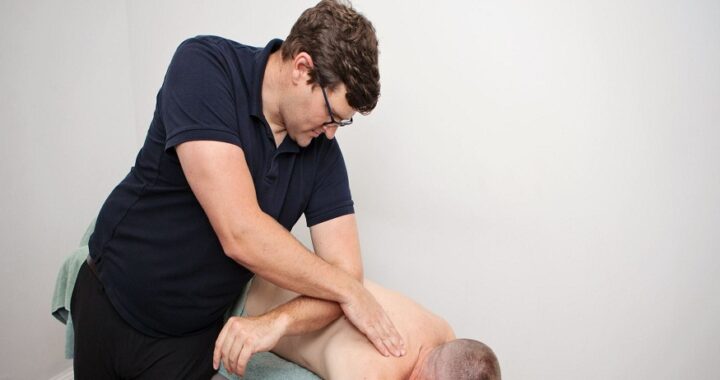 Why is remedial massage more expensive?
Why is remedial massage more expensive?  POST-SURGERY REHAB IN MERIDIAN: THE ROLE OF PHYSICAL THERAPY IN RECOVERY
POST-SURGERY REHAB IN MERIDIAN: THE ROLE OF PHYSICAL THERAPY IN RECOVERY  The Power Of Holistic Wellness: A Pathway To Mind-Body Harmony For Women
The Power Of Holistic Wellness: A Pathway To Mind-Body Harmony For Women  Enhancing Endurance and Recovery with Anavar: What You Should Know
Enhancing Endurance and Recovery with Anavar: What You Should Know 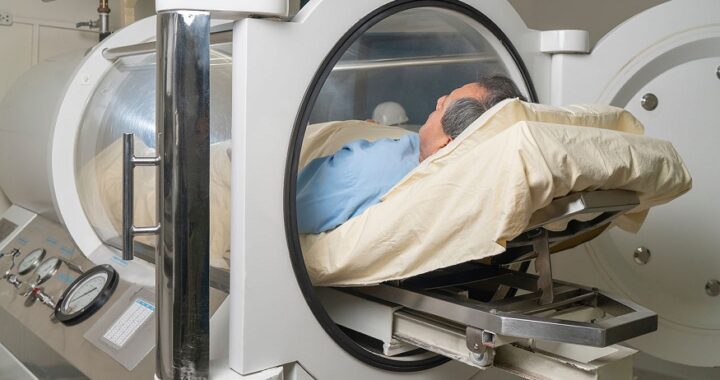 Hyperbaric Oxygen Therapy in Reproductive Medicine
Hyperbaric Oxygen Therapy in Reproductive Medicine